5 min read
In the late 1950s, thalidomide hit the market as a treatment for pregnant women with morning sickness. The drug had been tested on animals and deemed “safe” for human patients. Then hospitals began to fill with babies born without arms or legs, with severe organ damage, or not born alive at all. More than 10,000 babies were impacted in this medical failure that became known around the world as the “thalidomide tragedy”—and some of them still bear the devastating consequences to this day.
The thalidomide scandal shook the medical world and went down in history as one of the most catastrophic medical failures—but it wasn’t a fluke. It was a warning.

For decades, we’ve known that drugs routinely fail in clinical trials after animal tests have deemed them safe enough and sufficiently effective to give to human volunteers—nearly 95% of them. And as thalidomide showed us, some animal-tested drugs aren’t just ineffective in humans—they can be highly dangerous and even deadly. In some cases, after a drug has already harmed humans, experimenters have tested it on other animal species and discovered that some responded in ways similar to humans—but this hindsight underscores the need for human-relevant testing even more. That’s because there’s no reliable way to predict in advance which animals will react like humans.
While thalidomide may be the most infamous example of drug testing disasters, many other drugs have caused serious harm despite passing tests on animals. Here are just some examples.
The TGN1412 Disaster
In the early 2000s, six healthy volunteers participated in a clinical trial for TeGenero’s TGN1412—a drug that had been extensively tested on animals and was intended to treat leukemia and other diseases. But within 90 minutes of receiving the experimental drug, all six men were writhing in agony as their immune systems went into overdrive, leading to severe, life-threatening organ failure—reactions never seen in animal tests or earlier safety studies. With intensive medical care, they survived—but one was left with permanent damage.
The Poison Pill
Launched in 1999, Vioxx was aggressively marketed as a miracle solution for arthritis and chronic pain. But experiments on animals failed to reveal the deadly risk it posed to humans: heart attacks and strokes. The consequences were catastrophic, with rough estimates of up to 38,000 premature deaths and 88,000 heart attacks. Vioxx was pulled from the market in one of the largest voluntary drug recalls in history—but many patients who have taken the drug still suffer serious health complications.
The Fatal Fialuridine Failure
In 1993, 15 patients entered a clinical trial for fialuridine, a drug intended to treat hepatitis B that had passed tests in mice, rats, dogs, and monkeys. The trial was abruptly halted when one patient was hospitalized with liver failure. Even after the remaining participants were told to stop taking the drug, six more developed severe toxicity over the following weeks. Five patients died, and two survived only thanks to liver transplants.
Troglitazone
Troglitazone, a would-be treatment for type 2 diabetes, appeared safe in animal tests and lowered glucose, insulin, and lipids in mice, rats, and monkeys. But those tests couldn’t predict its deadly effects in humans. Within just three years on the U.S. market, at least two dozen human patients who took the drug suffered acute liver failure—some died, others needed life-saving liver transplants—causing the drug’s withdrawal in 2000.


Years later, researchers at the University of North Carolina revisited troglitazone using a computer program designed to predict how drugs affect the liver. One explained: “It turns out that animals do a poor job predicting human drug-induced liver injury. There are lots of explanations, but one important reason is that bile acids are different in each species.”
Let’s Keep History From Repeating Itself
Have we learned anything from the thalidomide and other tragedies? Today, pharmaceutical companies continue to conduct wasteful, unreliable tests on animals—even though humans and other animals have vastly different biology and physiology. How many more animals have to suffer in laboratories, dosed with experimental drugs or infected with deadly diseases—and how many humans will have to die or suffer permanent health problems—before we finally say “enough is enough”?
The Solution Science Needs
There’s a way we can spare both animals and humans—with animal-free, state-of-the-art technology. Organ-on-a-Chip systems are leading the way in drug testing: in a recent study, scientists tested 870 Liver-Chips to see how well they predicted drug-induced liver injury, a leading cause of drug failure. The results were clear—these chips outperformed tests on animals and other conventional models.
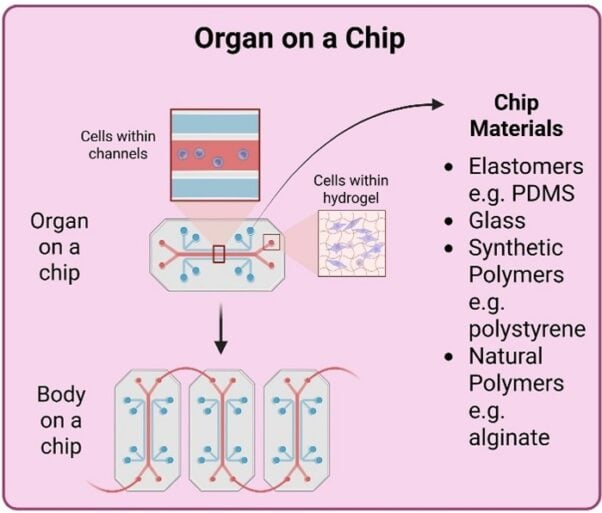

Other cutting-edge models are showing similar successes: the Blood Vessel-Chip has predicted blood clots caused by certain antibody drugs, and computer models of human heart cells can predict dangerous drug-induced heart rhythm problems with 89–96% accuracy, far outperforming tests on animals.
A study in 2018 found that advanced algorithms, using a large chemical database, can predict a new chemical’s toxicity better than tests on animals. One researcher said, “These results are a real eye-opener—they suggest that we can replace many animal tests with computer-based predictions and get more reliable results.”
These state-of-the-art methods point toward a future where fewer drugs fail in humans, fewer patients are harmed, and countless animals are spared from suffering.
Help Us Continue to Push Research Forward
There’s no better time than now. YOU can help us end cruel experiments on animals and push medicine forward by supporting PETA’s Research Modernization Now:








Discussion about this post